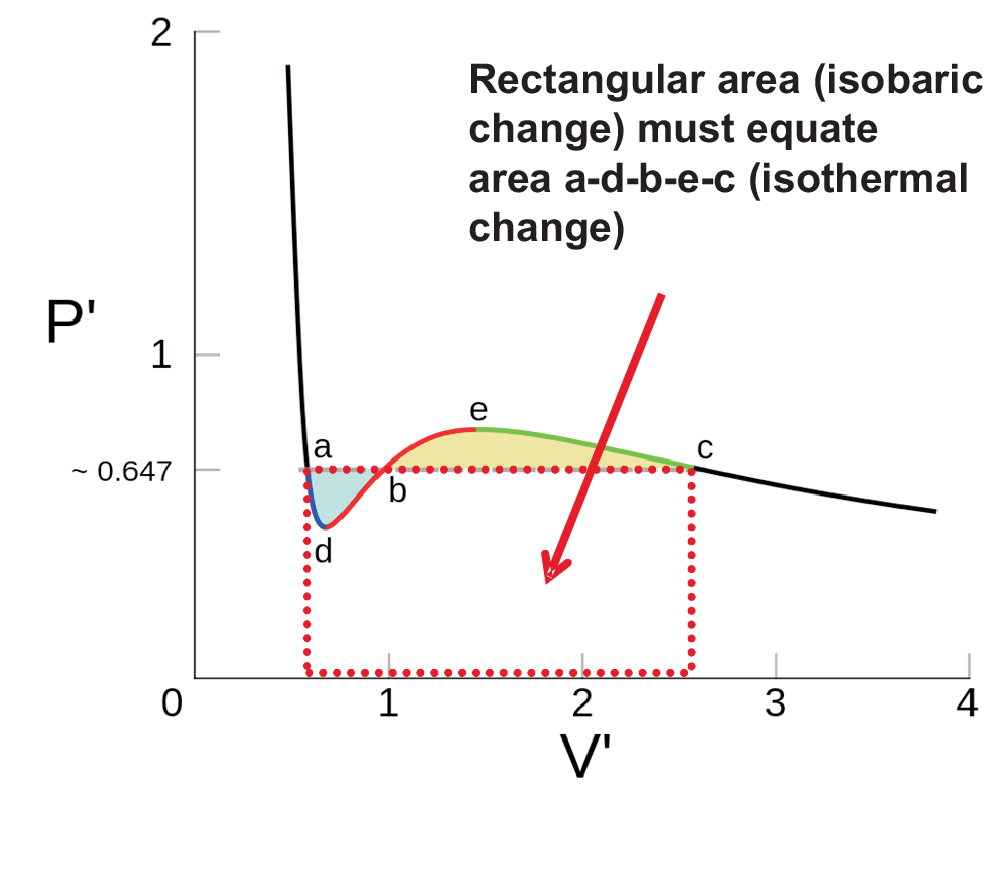
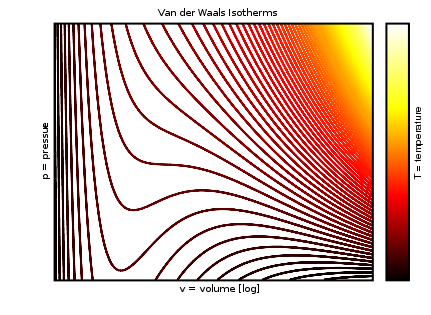
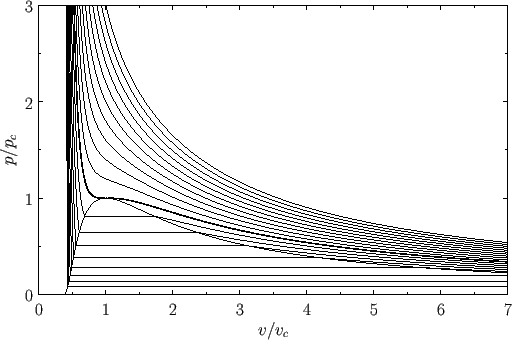
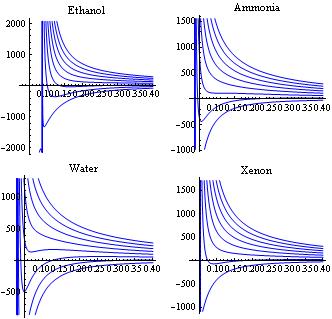
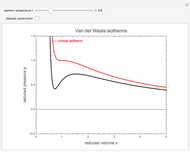
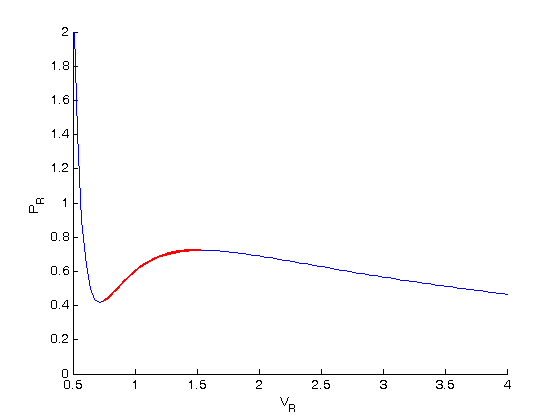
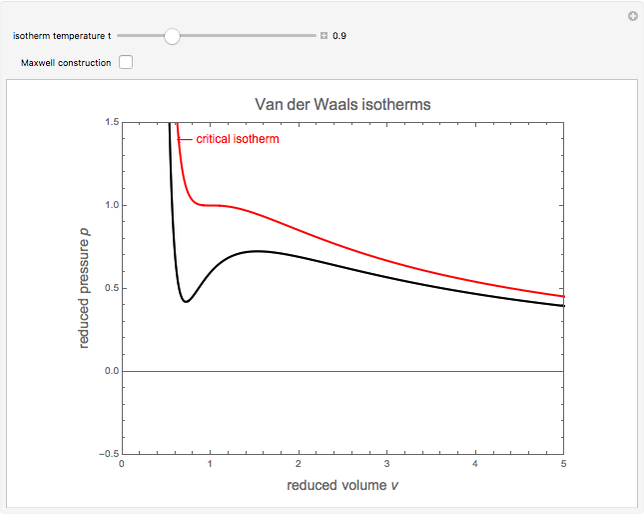
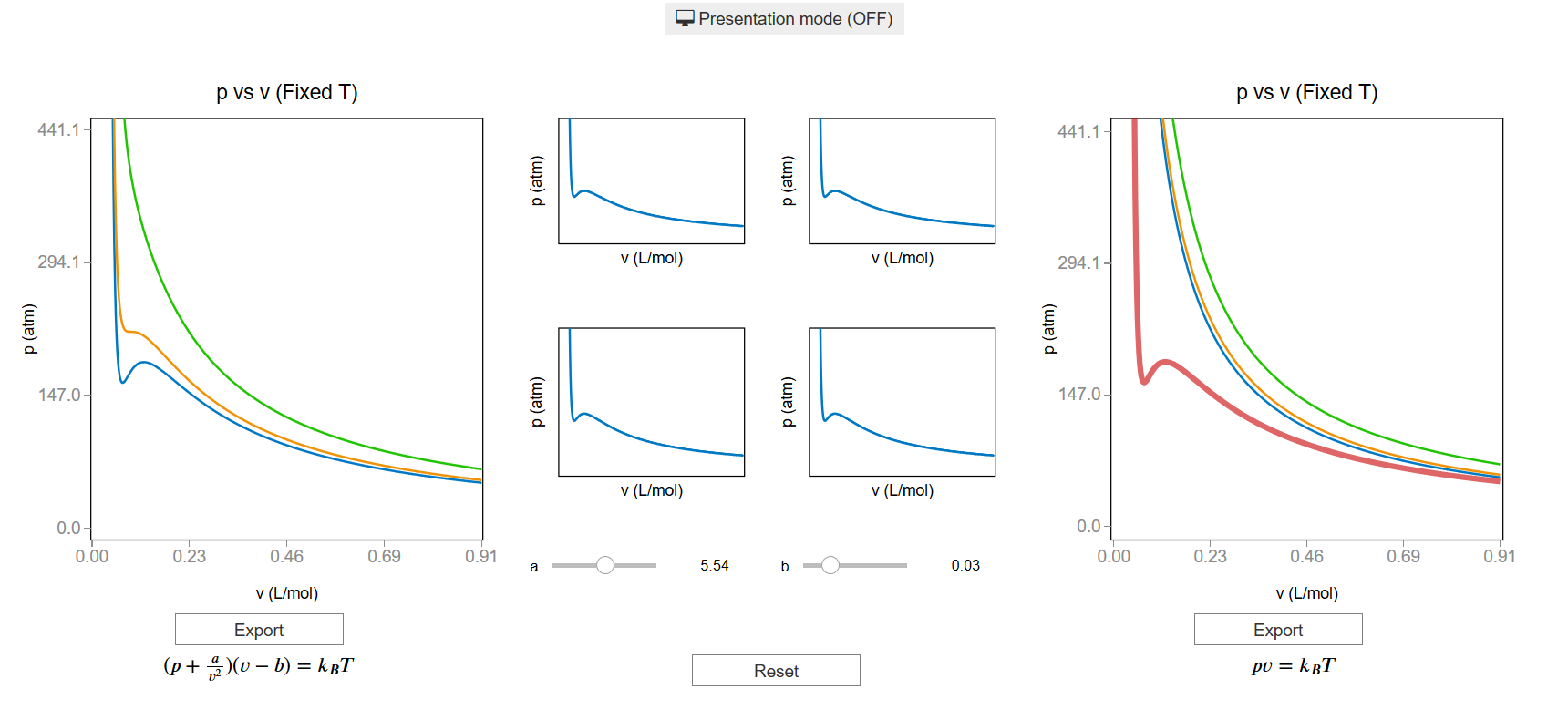
In van der Waals' gas equation, the pressure term is observed pressure plus intermolecular forces so that the pressure term becomes equal to the ideal pressure of the gas. But in the

homework and exercises - Doubts in Derivation of Critical constants, for real gases - Physics Stack Exchange
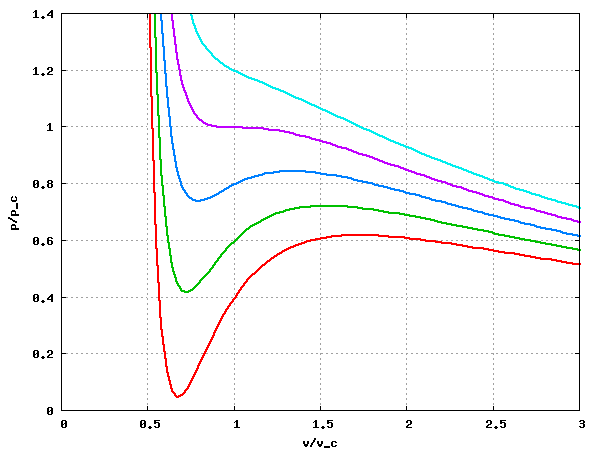
Van der Waals equation of state page on SklogWiki - a wiki for statistical mechanics and thermodynamics

Physics - Thermodynamics 2: Ch 32.1 Def. and Terms (20 of 25) van der Waals Equation Isotherms - YouTube