
Organismos Notificados: GMED (Francia) NB num. 0459 nuevo ON (ya 15) con MDR. Enhorabuena!!! | Red de Tecnologías Sanitarias y Productos Sanitarios
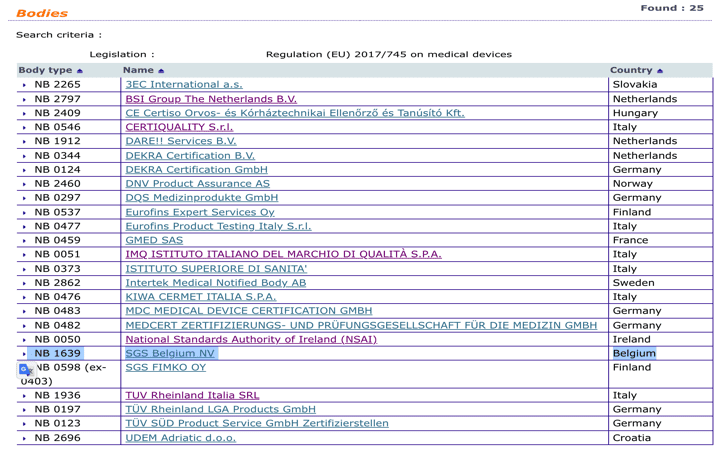
Organismos Notificados MDR (25): SGS Belgium (Belgica) ON num. 1639 nuevo ON. Enhorabuena @SGS_Spain !!! | Red de Tecnologías Sanitarias y Productos Sanitarios

Which EU Notified Bodies Have Been “Designated” Under the MDR 2017/745 and IVDR 2017/746? – Oriel STAT A MATRIX – ELIQUENT Life Sciences Blog

Neuronavigation, in Vitro Diagnostics, European Economic Community, transcranial Magnetic Stimulation, ISO 13485, cE Marking, european Commission, directive, medical Device, Regulation | Anyrgb













