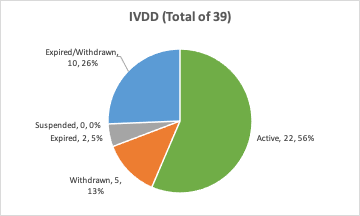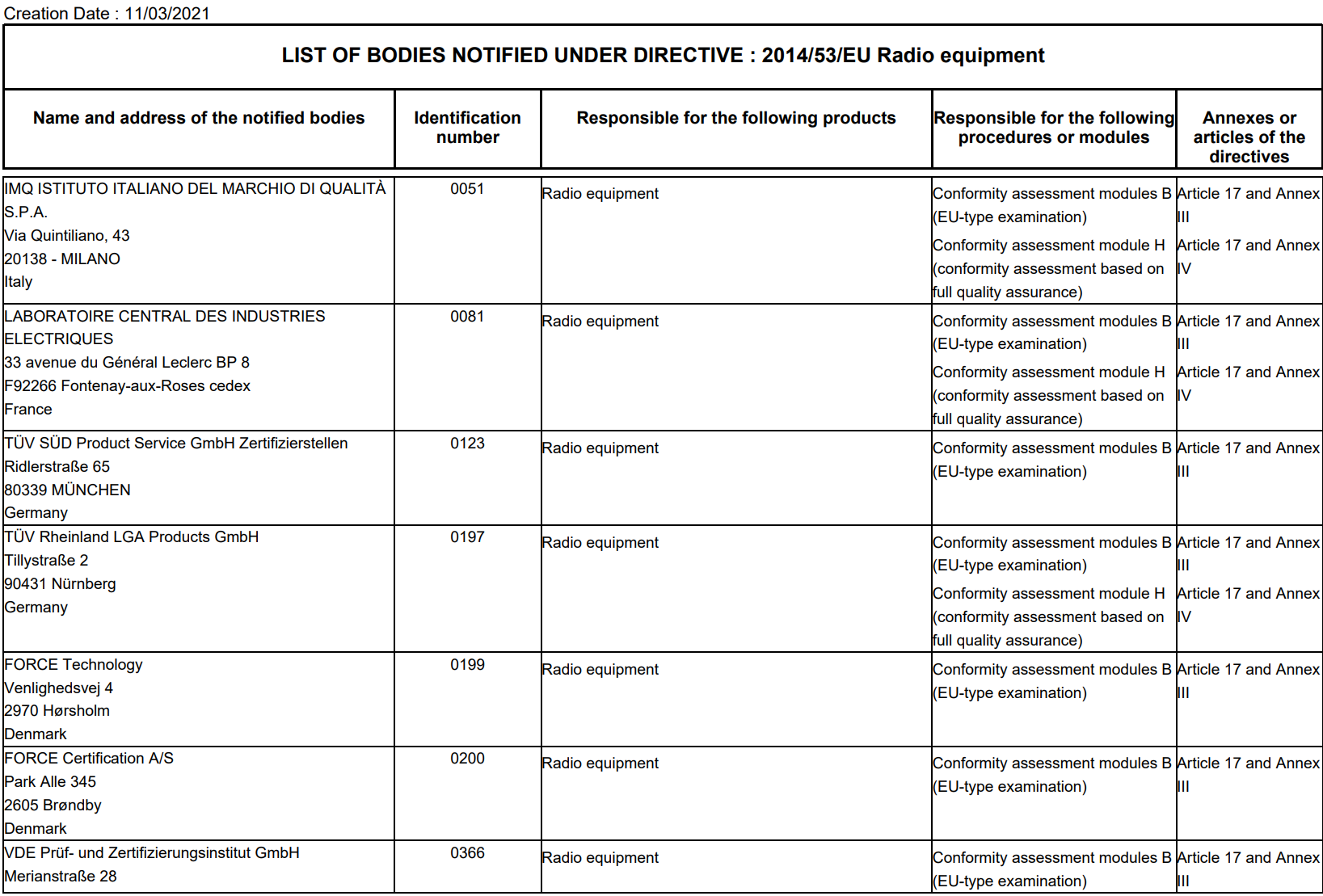
Organismos Notificados MDR (44): RISE – Medical Notified Body AB (Suecia) ON num. 3033 nuevo Organismo Notificado. Enhorabuena !!!
Organismos Notificados MDR (41): UDEM (Turquia) ON num. 2292 nuevo Organismo Notificado. Enhorabuena !!!
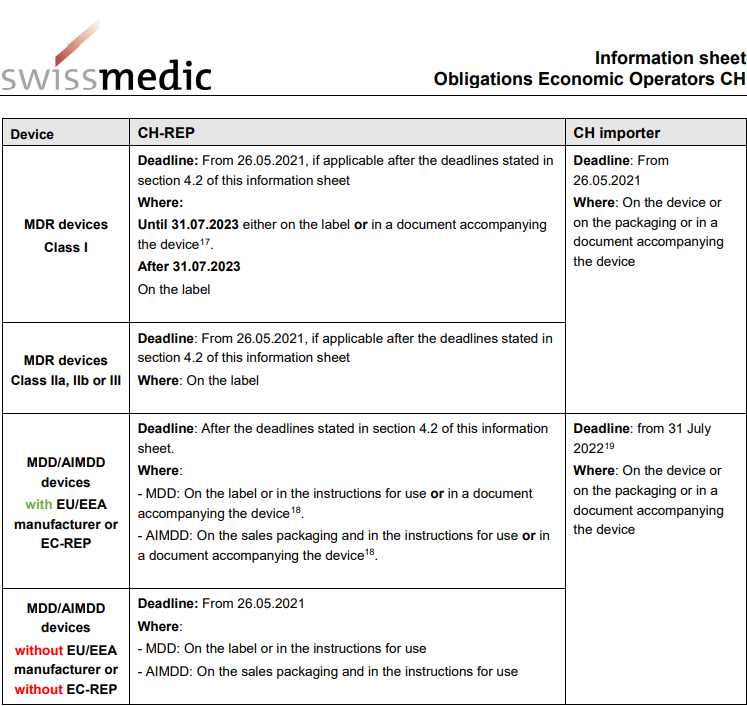
MDR: 26 Notified Bodies on NANDO & Swiss economic operator's requirements updated! · MDlaw – Information platform on European medical device regulations

Mario Gabrielli Cossellu on LinkedIn: #notifiedbody #mdr #nando #ivdr #conformityassessment #eu #regulations… | 11 comments

Organismos Notificados MDR (42): Scarlet NB BV (Países Bajos) ON num. 3022 nuevo Organismo Notificado. Enhorabuena !!! | Red de Tecnologías Sanitarias y Productos Sanitarios