How can a joint European health technology assessment provide an 'additional benefit' over the current standard of national assessments? : Insights generated from a multi-stakeholder survey in hematology/oncology. - Abstract - Europe
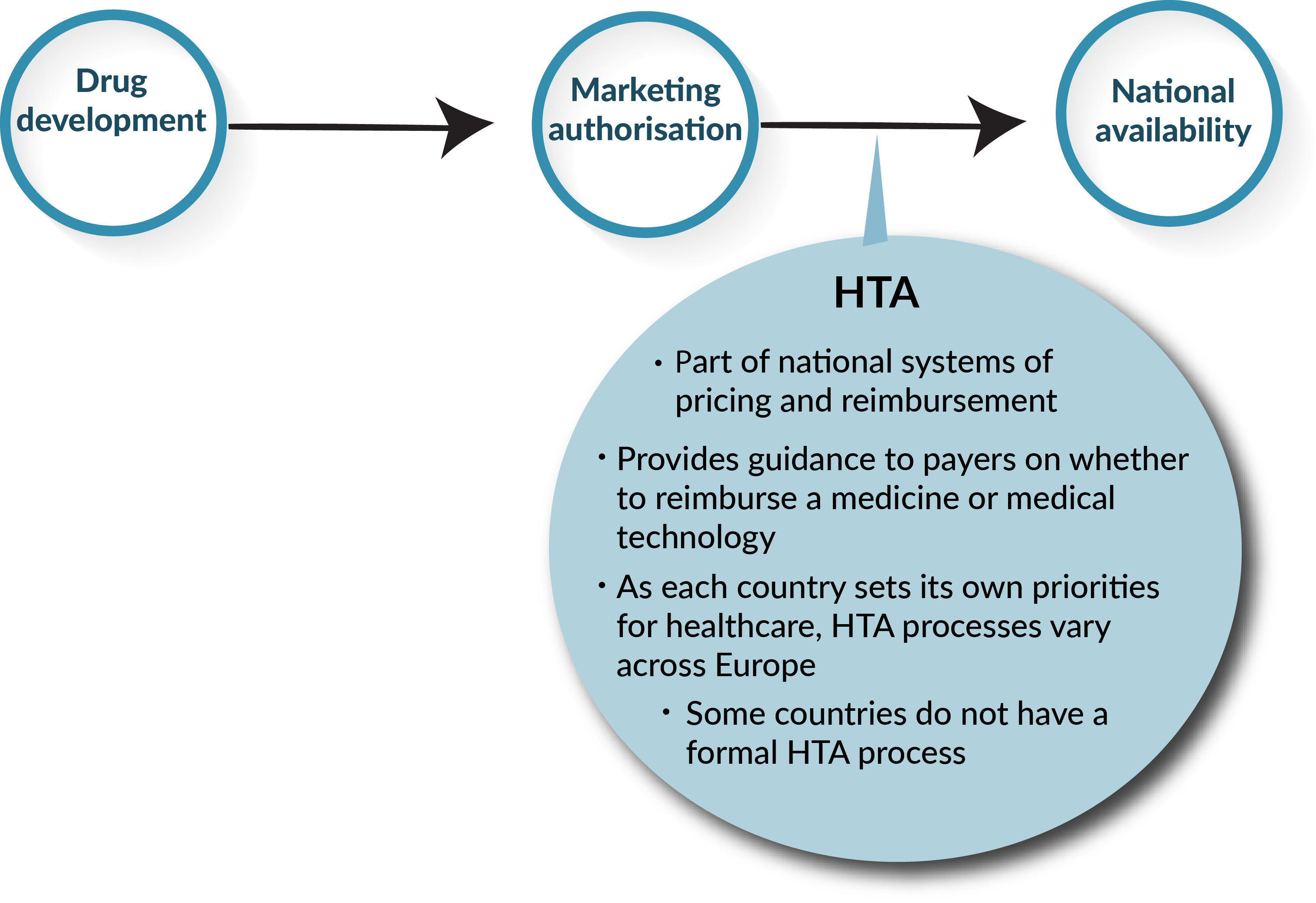
Early engagement with Health Technology Assessment authorities will accelerate product launch and improve chances for reimbursement

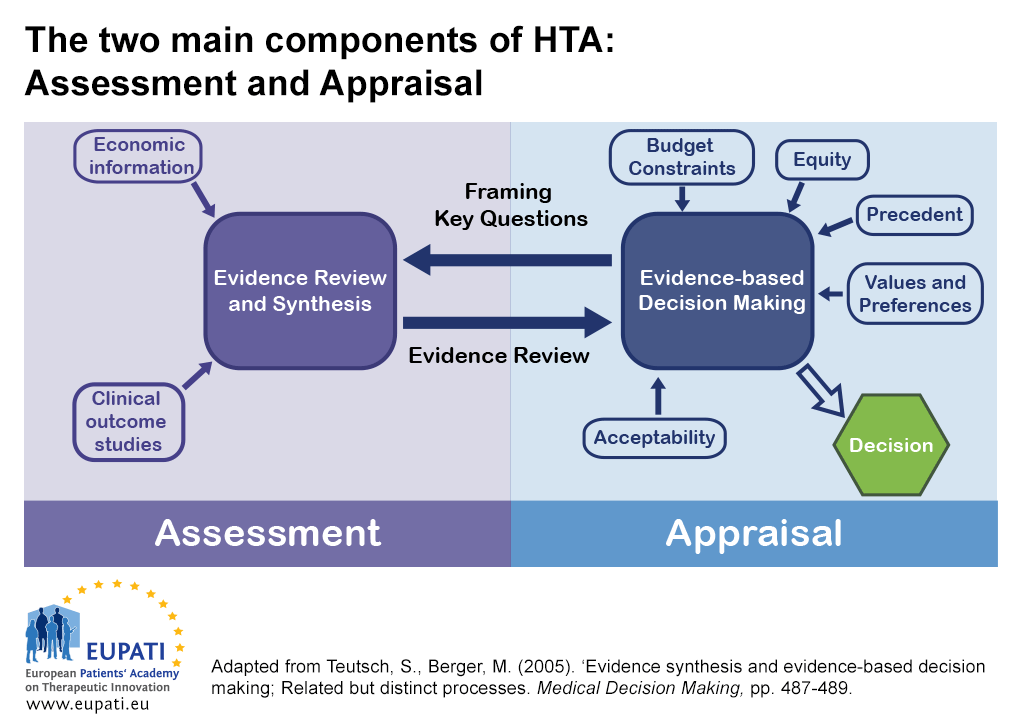
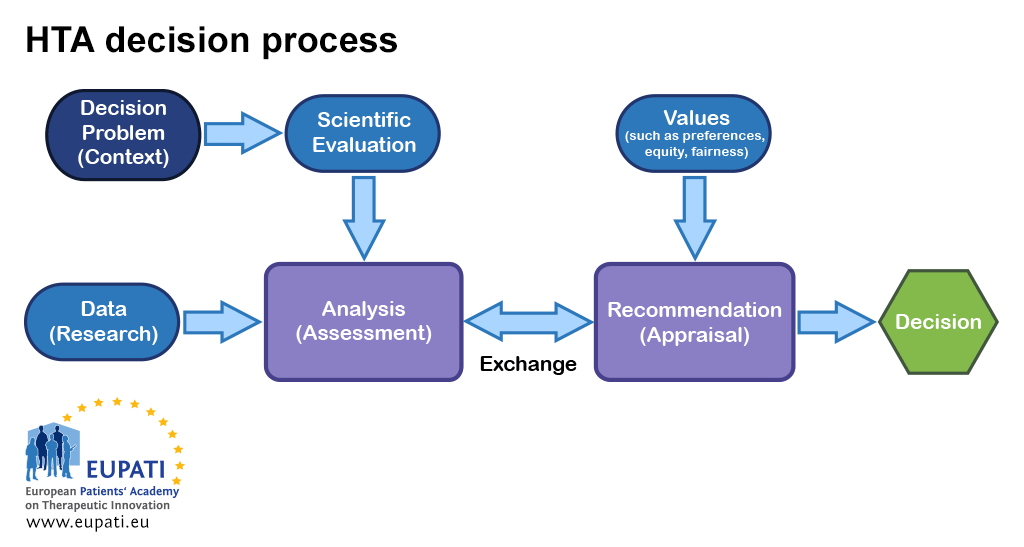
Full article: Differences in evidentiary requirements for oncology drug effectiveness assessments among six European health technology assessment bodies — can alignment be improved?

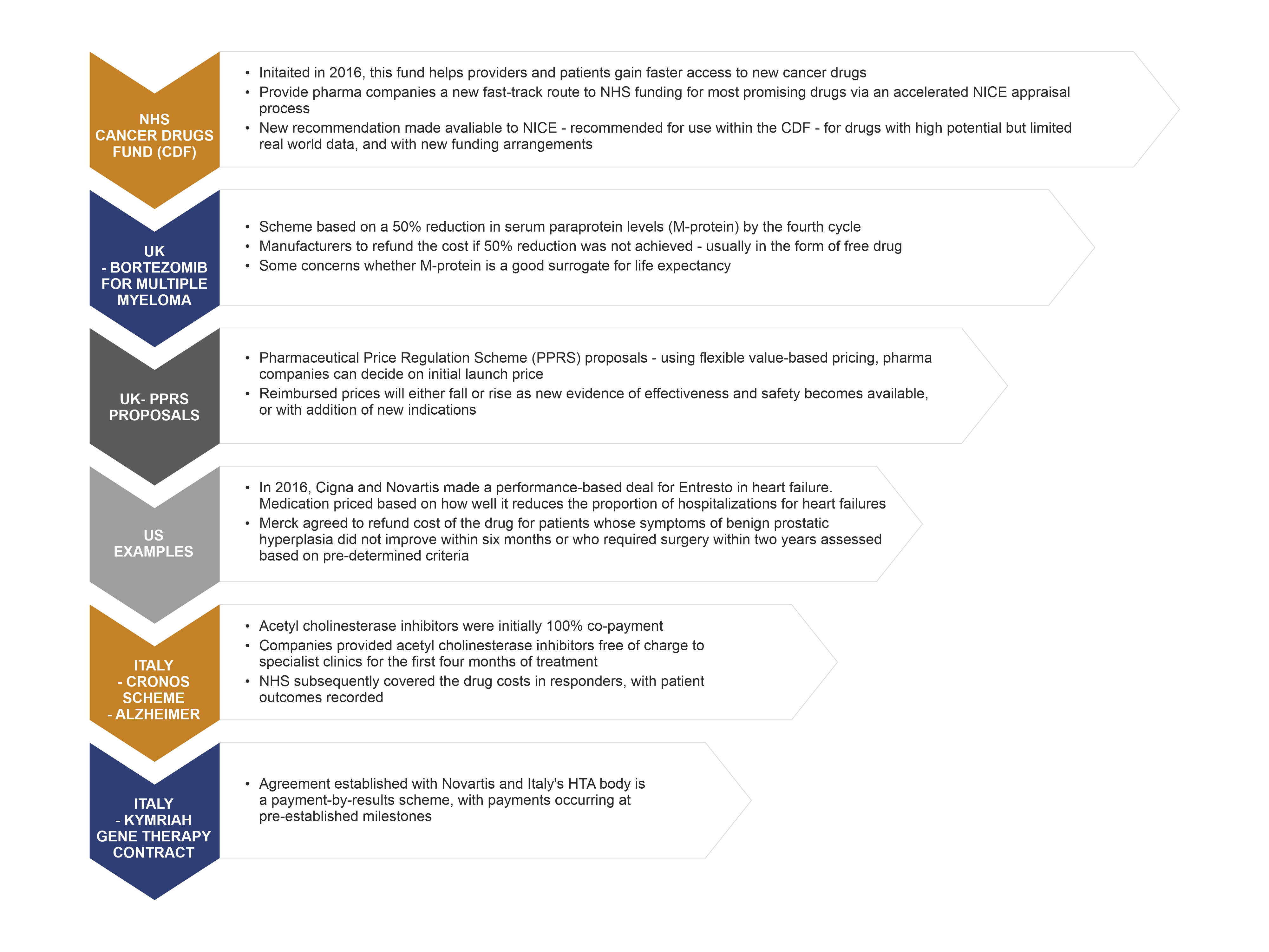
PDF) Weighing of Evidence by Health Technology Assessment Bodies: Retrospective Study of Reimbursement Recommendations for Conditionally Approved Drugs | Lourens T Bloem - Academia.edu
![PDF] Provisions and Special Considerations for Rare Diseases / Orphan Drugs by Health Technology Assessment (HTA) Bodies: Systematic Evaluation in 25 Countries | Semantic Scholar PDF] Provisions and Special Considerations for Rare Diseases / Orphan Drugs by Health Technology Assessment (HTA) Bodies: Systematic Evaluation in 25 Countries | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/14003250af4e607f52f1a5a0edead7f454917e31/1-Figure1-1.png)
PDF] Provisions and Special Considerations for Rare Diseases / Orphan Drugs by Health Technology Assessment (HTA) Bodies: Systematic Evaluation in 25 Countries | Semantic Scholar